A landmark ruling against two leading drug companies could save the NHS in England “hundreds of millions” a year.
Novartis and Bayer were trying to stop NHS doctors from prescribing a cheaper treatment for a serious eye condition.
Health bosses said the judgement in the High Court may reduce the power of companies to set prices.
Drug company Novartis said they were “disappointed” because patients were being asked to accept an unlicensed treatment to save the NHS money.
The case centred on the treatment of patients with the common eye condition, wet age-related macular degeneration (AMD).
Twelve NHS bodies in the north east of England were offering these patients Avastin, a cheaper alternative to the licensed drug, Lucentis.
David Hambleton, chief executive officer of NHS South Tyneside clinical commissioning group (CCG), one of the NHS groups involved in the case, welcomed the judgment, saying it was a good day for patients and the NHS.
“We’ve always said we think that it’s important that patients should have the choice of a very effective treatment for wet AMD, and it’s actually a fraction of the cost of the other alternatives.
“So I think what we do now is offer patients that choice. We believe that they will support very strongly having a cost-effective, safe treatment and saving the NHS generally a lot of money. It is a victory for common sense over commercial interests.”
What was the dispute?
The case was brought by pharmaceutical giants Bayer and Novartis against twelve NHS CCGs in the north of England.
The NHS groups were offering patients a choice between Lucentis and Eylea – drugs licensed for eye treatment – and the far cheaper drug Avastin, which is recommended by the World Health Organisation (WHO) for treating eyes, but only licensed for cancer treatment in the UK.
Novartis and Bayer manufacture the two more expensive licensed drugs – Lucentis which costs £561 and Eylea which costs £800.
By comparison, Avastin costs about £28 per injection.
Avastin is widely used around the world, particularly in the US.
In January, the National Institute for Health and Care Excellence (NICE) concluded that Avastin was as safe and effective as the two licensed drugs, Lucentis and Eylea.
What was the reaction?
Mike Burden, President of the Royal College of Ophthalmologists said: “Licensing laws are designed to protect patients from poorly regulated unproven drugs, but it is the drug companies’ responsibility to apply for a licence.
“We are treating 40,000 new diagnoses of wet AMD annually – the saving could amount to £500m a year. This amounts to one district hospital being built annually.”
David Hambleton, of NHS South Tyneside CCG, said the ruling would result in a rethink for NICE and the MHRA, the UK’s drug regulatory body.
“I think at least we’ve got some real legal clarity now, so both of the bodies – NICE and the MHRA – will need to look at what their guidance says.
“Now they have the option of allowing the use of so-called ‘off-label’ drugs.”
Responding to the ruling, a spokesperson from pharmaceutical company Novartis said: “Novartis is deeply disappointed in this decision and remains of the opinion that the policy undermines the well-established legal and regulatory framework that is there to protect both patients’ safety and to ensure health care professionals can prescribe with confidence.”
WHAT IS AGE-RELATED MACULAR DEGENERATION?
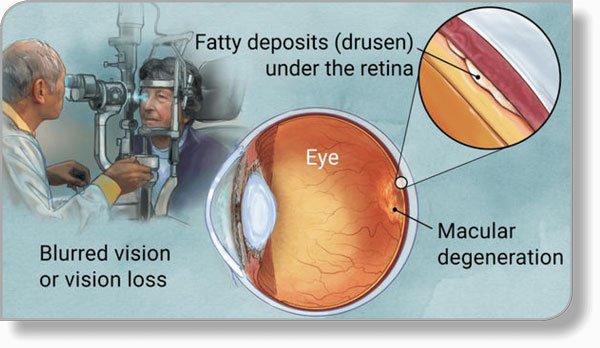
The most common form of AMD is “dry” AMD, which 90% of people experience. It’s the deterioration of the retinal pigment epithelial (tissue) cells within the macula. These cells support the light-sensitive photoreceptor cells that are so critical to vision. When we look at something, the photoreceptors, (a.k.a. rods and cones) collect the images and send them to the brain, where vision takes place.
The macula is an area near the center of the eye’s retina. This tiny area is responsible for our central, sharpest vision and determines our ability to read, drive a car, see faces or colors, and view objects in fine detail.
As AMD progresses, our central vision deteriorates and starts to fail. The presence of lipid (a fatty protein) deposits, called drusen, has also been connected with the disease. While drusen likely do not cause age-related macular degeneration (AMD), their existence within the eye increases a person’s risk of developing AMD.
In about 10 percent of patients, dry AMD can become “wet” AMD, where blood vessels form under the retina, leaking blood and fluid into the eye, according to AllAboutVision. Wet AMD further (and more quickly) distorts the central vision, and can ultimately lead to blindness.
SYMPTOMS: Distorted, blurry or spotty vision, inability to see clearly in dim lighting. Imagine if a straight line looks wavy!
RISK FACTORS: Aging is the major cause of AMD, and women are more likely to have the disease than men. Genetics also plays a role, as well as smoking, which can double your risk. AMD is more common in Caucasians than in African Americans and Hispanics, one study shows.
HOW YOU CAN IMPROVE YOUR EYE HEALTH: What Every Woman Should Know About Age-Related Macular Degeneration (AMD)
Add important nutrients called zeaxanthin and lutein into your diet to help support and improve your macular health. While these ingredients can be found in many leafy greens, oily fish and fruits, you most likely are not getting an adequate dose to significantly support and protect your macular health.
“The dietary carotenoids zeaxanthin and lutein protect the most important retinal real estate of the eye–-the macula–which allows us to see detail. It is therefore critical to maintain the quality and health of this area of retinal tissue in a modern society that depends upon using computer screens and driving automobiles, safely,” said Dr. Stuart Richer, OD, PhD
This is a "Pay as You Feel" website Please help keep us Ad Free.
You can have access to all of our online work for free. However if you want to support what we do, you could make a small donation to help us keep writing. The choice is entirely yours.












![[Full Clip] Starmer & Corbyn set out Labour’s position on the European Union (Withdrawal) Bill](https://nyebevannews.co.uk/wp-content/uploads/2017/09/corbyn-starmer-749064-100x70.jpg)










You must be logged in to post a comment.